Wholesale bulk API for compounding pharmacies.
Bulk active pharmaceutical ingredient supply with full chain-of-custody documentation. Built for 503A traditional compounding pharmacies operating under state board of pharmacy oversight.

What a pharmacy account includes.
Bulk API supply
USP-grade bulk active pharmaceutical ingredient designed for 503A compounding workflows.
Chain-of-custody documentation
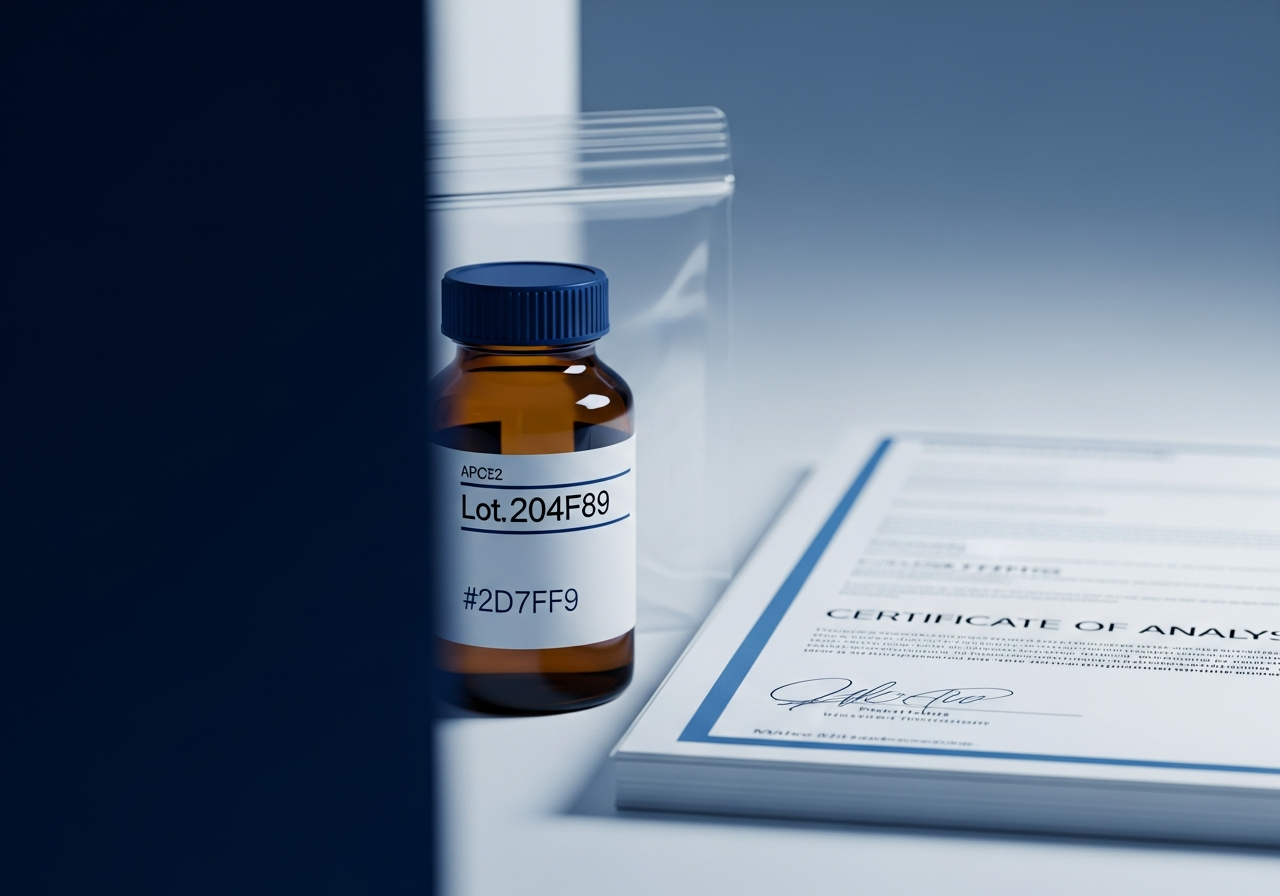
Full documentation packet: identity, HPLC purity, endotoxin testing, and supplier provenance.
Per-lot COA
Certificate of Analysis with every lot shipped, retained for your QA program.
Pharmacy-aware QA
Documentation packages built for state board of pharmacy inspection readiness.
Backup-supply planning
Multi-source contingency notes for high-demand APIs to keep your queue moving.
Account management
One U.S. point of contact for sourcing, documentation, and supply planning.
Common pharmacy questions.
State board of pharmacy license, DEA where applicable, NPI, pharmacist-in-charge contact, and a brief facility profile.
For 503A traditional compounding, we supply bulk API with full chain-of-custody documentation. We do not supply finished-dose 503B product to pharmacies.
Eligibility varies by API and current FDA list status. We provide documentation per item; pharmacies are responsible for verifying state-board and FDA-list eligibility before compounding.
No. We ship to U.S. licensed pharmacies only.
Most pharmacy accounts are verified within 1–2 business days of receiving complete documentation.